The Vaccine Works Fast
From the FDA report:
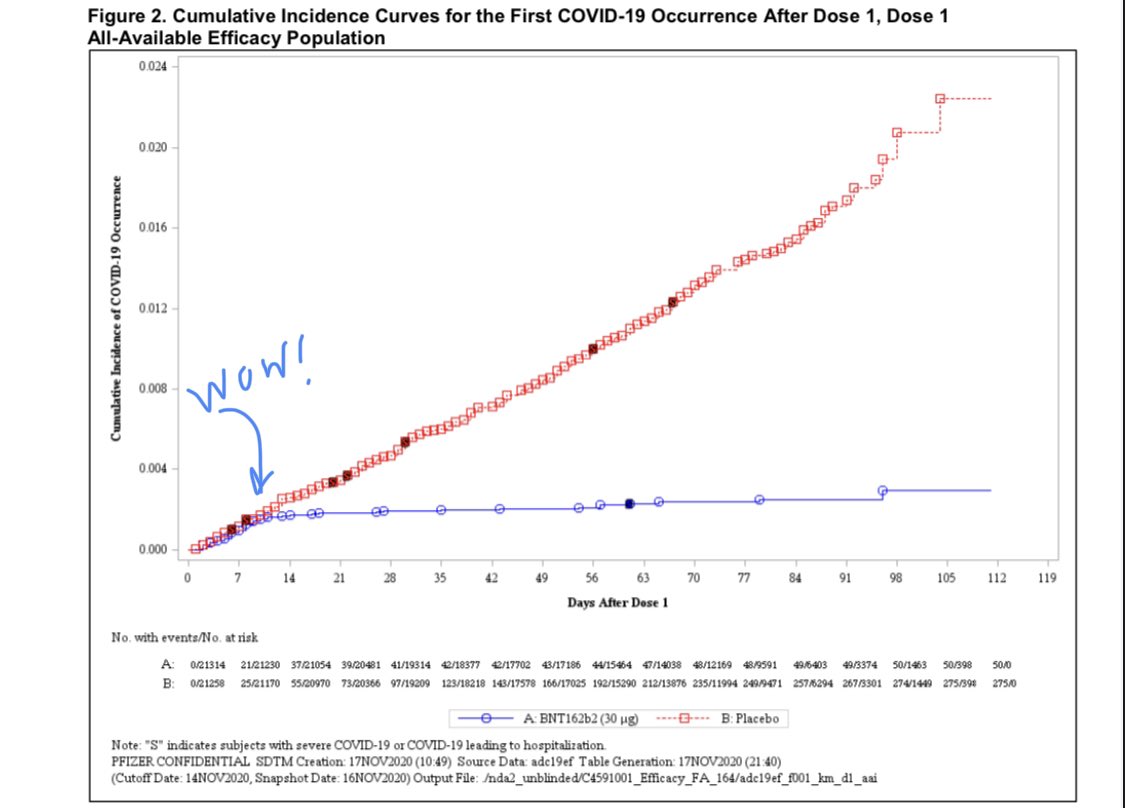
Two doses is great but one dose already looks good even though sample is small. “Efficacy against severe COVID-19 occurring after the first dose was 88.9%.” (Added. n.b this is over the entire sample.)
Based on the number of cases accumulated after Dose 1 and before Dose 2, there does seem to be some protection against COVID-19 disease following one
dose; however, these data do not provide information about longer term protection beyond 21 days after a single dose.
This is potentially important as we could vaccinate more people in a hot-spot and potentially delay the second dose. Noting, however, that this is a post-hoc analyses and the second dose came within 3 weeks.
As expected, a bit of pain, swelling and fatigue in a minority of participants are the biggest issues. No major safety concerns. Of course, we still need to monitor long-term.
Serious adverse events, while uncommon (<1.0%), represented medical events that occur in the general population at similar frequency as observed in the study…. No specific safety concerns were identified in subgroup analyses by age, race, ethnicity, medical comorbidities, or prior SARS-CoV-2 infection.
Hat tip: Biostatistician LucyStats.