Category: Law
AI and the FDA
Dean Ball has an excellent survey of the AI landscape and policy that includes this:
The speed of drug development will increase within a few years, and we will see headlines along the lines of “10 New Computationally Validated Drugs Discovered by One Company This Week,” probably toward the last quarter of the decade. But no American will feel those benefits, because the Food and Drug Administration’s approval backlog will be at record highs. A prominent, Silicon Valley-based pharmaceutical startup will threaten to move to a friendlier jurisdiction such as the United Arab Emirates, and they may in fact do it.
Eventually, I expect the FDA and other regulators to do something to break the logjam. It is likely to perceived as reckless by many, including virtually everyone in the opposite party of whomever holds the White House at the time it happens. What medicines you consume could take on a techno-political valence.
Agreed—but the nearer-term upside is repurposing. Once a drug has been FDA approved for one use, physicians can prescribe it for any use. New uses for old drugs are often discovered, so the off-label market is large. The key advantage of off-label prescribing is speed: a new use can be described in the medical literature and physicians can start applying that knowledge immediately, without the cost and delay of new FDA trials. When the RECOVERY trial provided evidence that an already-approved drug, dexamethasone, was effective against some stages of COVID, for example, physicians started prescribing it within hours. If dexamethasone had had to go through new FDA-efficacy trials a million people would likely have died in the interim. With thousands of already approved drugs there is a significant opportunity for AI to discover new uses for old drugs. Remember, every side-effect is potentially a main effect for a different condition.
On Ball’s main point, I agree: there is considerable room for AI-discovered drugs, and this will strain the current FDA system. The challenge is threefold.
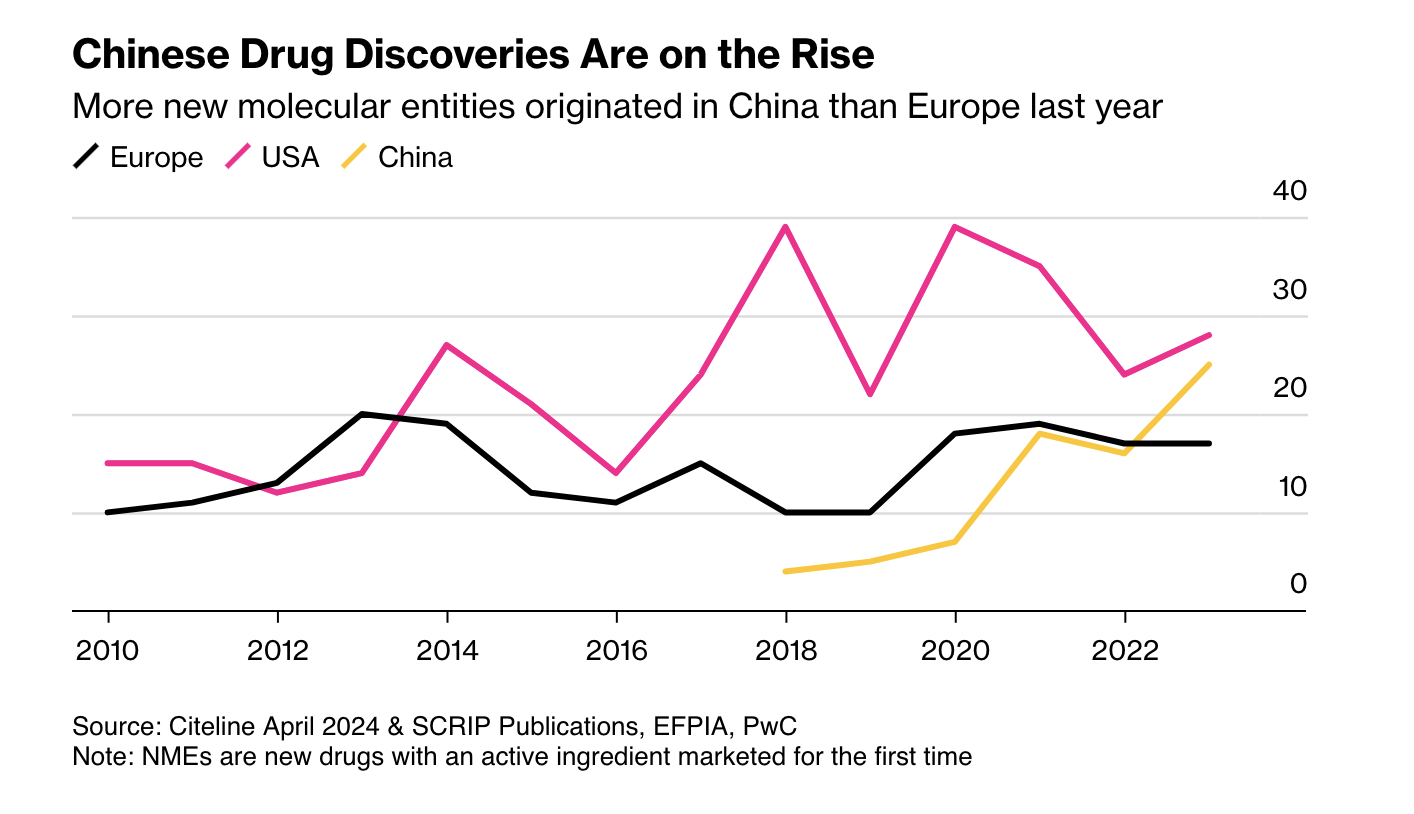
First, as Ball notes, more candidate drugs at lower cost means other regulators may become competitive with the FDA. China is the obvious case: it is now large and wealthy enough to be an independent market, and its regulators have streamlined approvals and improved clinical trials. More new drugs now emerge from China than from Europe.
Second, AI pushes us toward rational drug design. RCTs were a major advance, but they are in some sense primitive. Once a mechanic has diagnosed a problem, the mechanic doesn’t run a RCT to determine the solution. The mechanic fixes the problem! As our knowledge of the body grows, medicine should look more like car repair: precise, targeted, and not reliant on averages.
Closely related is the rise of personalized medicine. As I wrote in A New FDA for the Age of Personalized, Molecular Medicine:
Each patient is a unique, dynamic system and at the molecular level diseases are heterogeneous even when symptoms are not. In just the last few years we have expanded breast cancer into first four and now ten different types of cancer and the subdivision is likely to continue as knowledge expands. Match heterogeneous patients against heterogeneous diseases and the result is a high dimension system that cannot be well navigated with expensive, randomized controlled trials. As a result, the FDA ends up throwing out many drugs that could do good.
RCTs tell us about average treatment effects, but the more we treat patients as unique, the less relevant those averages become.
AI holds a lot of promise for more effective, better targeted drugs but the full promise will only be unlocked if the FDA also adapts.
Michael Clemens on H1-B visas
From 1990 to 2010, rising numbers of H-1B holders caused 30–50 percent of all productivity growth in the US economy. This means that the jobs and wages of most Americans depend in some measure on these workers.
The specialized workers who enter on this visa fuel high-tech, high-growth sectors of the 21st century economy with skills like computer programming, engineering, medicine, basic science, and financial analysis. Growth in those sectors sparks demand for construction, food services, child care, and a constellation of other goods and services. That creates employment opportunities for native workers in all sectors and at all levels of education.
This is not from a textbook narrative or a computer model. It is what happened in the real world following past, large changes in H-1B visa restrictions. For example, Congress tripled the annual limit on H-1B visas after 1998, then slashed it by 56 percent after 2004. That produced large, sudden shocks to the number of these workers in some US cities relative to others. Economists traced what happened to various economic indicators in the most-affected cities versus the least-affected but otherwise similar cities. The best research exhaustively ruled out other, confounding forces.
That’s how we know that workers on H-1B visas cause dynamism and opportunity for natives. They cause more patenting of new inventions, ideas that create new products and even new industries. They cause entrepreneurs to found more (and more successful) high-growth startup firms. The resulting productivity growth causes more higher-paying jobs for native workers, both with and without a college education, across all sectors. American firms able to hire more H-1B workers grow more, generating far more jobs inside and outside the firm than the foreign workers take.
An important, rigorous new study found the firms that win a government lottery allowing them to hire H-1B workers produce 27 percent more than otherwise-identical firms that don’t win, employing more immigrants but no fewer US natives—thus expanding the economy outside their own walls. So, when an influx of H-1B workers raised a US city’s share of foreign tech workers by 1 percentage point during 1990–2010, that caused7 percent to 8 percent higher wages for college-educated workers and 3 percent to 4 percent higher wages for workers without any college education.
Here is the full piece.
H1-B visa fees and the academic job market
Assume the courts do not strike this down (perhaps they will?).
Will foreigners still be hired at the entry level with an extra 100k surcharge? I would think not,as university budgets are tight these days. I presume there is some way to turn them down legally, without courting discrimination lawsuits?
What if you ask them to accept a lower starting wage? A different deal in some other manner, such as no summer money or a higher teaching load? Is that legal? Will schools have the stomach to even try? I would guess not. Is there a way to amortize the 100k over five or six years? What if the new hire leaves the institution in year three of the deal?
In economics at least, a pretty high percentage of the graduate students at top institutions do not have green cards or citizenships.
So how exactly is this going to work? There are not so many jobs in Europe, not enough to absorb those students even if they wish to work there. Will many drop out right now? And if the flow of graduate students is not replenished, given that entry into the US job market is now tougher, how many graduate programs will close up?
Will Chinese universities suddenly hire a lot more quality talent?
Here is some related discussion on Twitter.
As they say, solve for the equilibrium…
My excellent Conversation with David Commins
Saudi Arabia and the Gulf are the topics, here is the audio, video, and transcript. Here is the episode summary:
David Commins, author of the new book Saudi Arabia: A Modern History, brings decades of scholarship and firsthand experience to explain the kingdom’s unlikely rise. Tyler and David discuss why Wahhabism was essential for Saudi state-building, the treatment of Shiites in the Eastern Province and whether discrimination has truly ended, why the Saudi state emerged from its poorer and least cosmopolitan regions, the lasting significance of the 1979 Grand Mosque seizure by millenarian extremists, what’s kept Gulf states stable, the differing motivations behind Saudi sports investments, the disappointing performance of King Abdullah University of Science and Technology despite its $10 billion endowment, the main barrier to improving its k-12 education, how Yemen became the region’s outlier of instability and whether Saudi Arabia learned from its mistakes there, the Houthis’ unclear strategic goals, the prospects for the kingdom’s post-oil future, the topic of David’s next book, and more.
And an excerpt:
COWEN: Now, as you know, the senior religious establishment is largely Nejd, right? Why does that matter? What’s the historical significance of that?
COMMINS: Right. Nejd is the region of central Arabia. Riyadh is currently the capital. The first Saudi empire had a capital nearby, called Diriyah. Nejd is really the territory that gave birth to the Wahhabi movement, it’s the homeland of the Saud dynasty, and it is the region of Arabia that was most thoroughly purged of the older Sunni tradition that had persisted in Nejd for centuries.
Consequently, by the time that the Saudi government developed bureaucratic agencies in the 1950s and ’60s, the religious institution was going to recruit from that region of Arabia primarily. Now, it certainly attracted loyalists from other parts of Arabia, but the Wahhabi mission, as I call it — their calling to what they considered true belief — began in Nejd and was very strongly identified with the towns of Nejd ever since the late 1700s.
COWEN: Would I be correct in inferring that some of the least cosmopolitan parts of Saudi Arabia built the Saudi state?
COMMINS: Yes, that is correct. That is correct. If you think of the 1700s and 1800s, the Red Sea and Persian Gulf coast of Arabia were the most cosmopolitan parts of Arabia.
COWEN: They’re richer, too, right? Jeddah is a much more advanced city than Riyadh at the time.
COMMINS: Somewhat more advanced. Yes, it is more advanced, it is more cosmopolitan than Nejd. There is the regional identity in Hejaz, that is the Red Sea coast where the holy cities and Jeddah are located. The townspeople there tended to look upon Nejd as a less advanced part of Arabia. But again, that’s a very recent historical development.
COWEN: How is it that the coastal regions just dropped the ball? You could imagine some alternate history where they become the center of Saudi power and religious thought, but they’re not.
COMMINS: Right. If you take Jeddah, Mecca, and Medina — that region of Arabia, known as Hejaz, had always been under the rule of other Muslim empires. They were under the rule of other Muslim powers because of the religious value of possessing, if you will, the holy cities, Mecca and Medina. From the time of the first Muslim dynasty that was based in Damascus in the seventh and early eighth centuries, all the way until the Ottoman Empire, Muslim dynasties outside Arabia coveted control of that region. They were just more powerful than local resources could generate.
Hejaz was always, if you were, to dependency on outside Muslim powers. If you look at the east coast of Arabia — what’s now the Eastern Province of Saudi Arabia and the Persian Gulf — it was richer than central Arabia. It’s the largest oasis in Arabia. It is in proximity to pearling banks, which were an important source for income for residents there. It was part of the Indian Ocean trade between Iraq and India. The population there was always — well, always — for the last thousand years has been dominated by Bedouin tribesmen.
There was a brief Ismaili Shia republic, you might say, in that part of Arabia in medieval times. It just didn’t have, it seems, the cohesion to conquer other parts of Arabia. That’s what makes the Saudi story really remarkable, is that they were able to muster and sustain the cohesion to carry out a conquest like that over the course of 50 years.
COWEN: Physically, how did they manage that? Water is a problem, a lot of transport is by camel, there’s no real rail system, right?
Recommended, full of historical information about a generally neglected region, neglected from the point of view of history at least rather than current affairs.
Summary of a new DeepMind paper
Super intriguing idea in this new @GoogleDeepMind paper – shows how to handle the rise of AI agents acting as independent players in the economy.
It says that if left unchecked, these agents will create their own economy that connects directly to the human one, which could bring both benefits and risks.
The authors suggest building a “sandbox economy,” which is a controlled space where agents can trade and coordinate without causing harm to the broader human economy.
A big focus is on permeability, which means how open or closed this sandbox is to the outside world. A fully open system risks crashes and instability spilling into the human economy, while a fully closed system may be safer but less useful.
They propose using auctions where agents fairly bid for resources like data, compute, or tools. Giving all agents equal starting budgets could help balance power and prevent unfair advantages.
For larger goals, they suggest mission economies, where many agents coordinate toward one shared outcome, such as solving a scientific or social problem.
The risks they flag include very fast agent negotiations that humans cannot keep up with, scams or prompt attacks against agents, and powerful groups dominating resources.
To reduce these risks, they call for identity and reputation systems using tools like digital credentials, proof of personhood, zero-knowledge proofs, and real-time audit trails.
The core message is that we should design the rules for these agent markets now, so they grow in a safe and fair way instead of by accident.
That is from Rohan Paul, though the paper is by Nenad Tomasev, et.al. It would be a shame if economists neglected what is perhaps the most important (and interesting) mechanism design problem facing us.
Should we abolish mandatory quarterly corporate reporting?
President Trump has suggested doing that. I have not found a human source as good as GPT5, so I will cite that:
Theory predicts that more frequent reporting can exacerbate managerial short‑termism; some archival evidence finds lower investment when reporting frequency rises. But when countries reduced frequency (UK/EU), the average firm’s investment didn’t materially change—in part because most issuers kept giving quarterly updates anyway…
Will markets just insist on quarterly anyway? That’s what happened in the UK and Austria: after rules allowed semi‑annual reporting, only a small minority actually stopped quarterly updates; those that did often saw lower liquidity and less analyst coverage. So yes—many issuers kept some form of quarterly communication to satisfy investors.
There is much, much more at the link.
Intertemporal substitution
Across several Central American nations money transfers have jumped 20 percent.
The reason, officials, migrants and analysts say, is that people afraid of being deported are trying to get as much money out of the country as possible, while they still can.
The money transfers, called remittances, are a critical lifeline for many countries and families around the world, especially in Central America and the Caribbean. There, the funds sometimes make up a huge chunk of a nation’s economy — as much as a quarter of a country’s gross domestic product, as in Honduras and Nicaragua.
Here is more from James Wagner at the NYT.
What should I ask Cass Sunstein?
Yes, I will be doing a Conversation with him soon. Most of all (but not exclusively) about his three recent books Liberalism: In Defense of Freedom, Manipulation: What It Is, Why It Is Bad, What To Do About It, and Imperfect Oracle: What AI Can and Cannot Do.
So what should I ask him? Here is my previous CWT with Cass.
The British War on Slavery
In August of 1833 the British passed legislation abolishing slavery within the British Empire and putting more than 800,000 enslaved Africans on the path to freedom. To make this possible, the British government paid a huge sum, £20 million or about 5% of GDP at the time, to compensate/bribe the slaveowners into accepting the deal. In inflation adjusted terms this is about £2.5 billion today (2025) but relative to GDP the British spent an equivalent of about $170 billion to free the slaves, a very large expenditure.

Indeed, the expenditure was so large that the money was borrowed and the final payments on the debt were not made until 2015. When in 2015 a tweet from the British Treasury revealed this surprising fact, there was a paroxysm of outrage as if slaveholders were still being paid off. I see the compensation in much more positive terms.
Of course, in an ideal world, compensation would have been paid to the slaves, not the slaveowners. Every man has a property in his own person and it was the slaves who had had their property stolen. In an ideal world, however, slavery would never have happened. Thus, the question the British abolitionists faced is not what happens in an ideal world but how do we get from where we are to a better world? Compensating the slaveowners was the only practical and peaceful way to get to a better world. As the great abolitionist William Wilberforce said on his deathbed “Thank God that I should have lived to witness a day in which England is willing to give twenty millions sterling for the abolition of slavery!”
The 1833 Slavery Abolition Act was preceded by the 1807 Slave Trade Act which had banned trade in slaves. In an excellent new paper, The long campaign: Britain’s fight to end the slave trade, economist historians Yi Jie Gwee and Hui Ren Tan assemble new archival data to assess how the Royal Navy’s anti slave-trade patrols expanded over time, how effective they were at curtailing the trade, the influence of supply-side enforcement versus demand-side changes on ending the trade, and why Britain persisted with this costly campaign.
Britain’s naval suppression campaign began on a modest scale but the campaign grew in strength throughout the 19th century, peaking in the late 1840s to early 1850s when over 14% of the entire Royal Navy fleet was deployed to anti-slavery patrols. The British patrols captured some 1,600 ships and freed some 150,000 people destined for slavery but they were at best only modestly successful at reducing the slave trade. The big impact came when Brazil, the largest remaining market for enslaved labor (by the mid-19th century, nearly 80% of trans-Atlantic slave voyages sailed under Brazilian or Portuguese flags), enacted its anti slave-trade law in 1850. Britain’s campaign was not without influence on the demand side however as passage of the Aberdeen Act in 1845 allowed the Royal Navy to seize Brazilian slave ships and that put pressure on Brazil and helped spur the 1850 law.
The suppression patrols were expensive (consuming ships, men, and funds), and Britain derived no direct economic benefit from them. Yet, even during the Napoleonic Wars, the Opium Wars, and the Crimean War, the Royal Navy continued to station ships, even high-tech steam ships, off West Africa to catch slavers. Due to the expense, the patrols were controversial and there were attempts to end them. Gwee and Tan look at the votes on ending the patrols and find that ideology was the dominant factor explaining support for the patrols, that is a principled opposition to the slave trade and a belief in the moral cause of abolition kept Britain in the war against slavery even at considerable expense.
Ordinarily, I teach politics without romance and look for interest as an explanation of political action and while I don’t doubt that doing good and doing well were correlated, even during abolition, I also agree with Gwee and Tan that the British war on slavery was primarily driven by ideology and moral principle as both the compensation plan and the support of the anti-slavery patrols attest.
British taxpayers shouldered an enormous military and financial burden to eliminate slavery, reflecting a generosity of spirit and a sincere attempt to address a moral wrong—an act of atonement that stands as one of the most unusual and significant in history.
What the financial regulators are saying and feeling
1. “Yes, we know stablecoins will have one hundred percent reserves, but we are not sure we can regulate that system into a position of safety.”
2. “Well, the rest of the financial system has nothing like one hundred percent reserves, but don’t worry we have everything there under control.”
The hole is large enough to drive a truck through. Keep this contrast in mind, because you will be hearing it, expressed in other terms of course, hundreds of times over the next year or so.
They solved for the Kansas City Chiefs enforcement equilibrium
We examine how financial pressure influences rule enforcement by leveraging a novel setting: NFL officiating. Unlike traditional regulatory environments, NFL officiating decisions are immediate, transparent, and publicly scrutinized, providing a unique empirical lens to test whether a worsening financial climate shapes enforcement behavior. Analyzing 13,136 defensive penalties from 2015 to 2023, we find that postseason officiating disproportionately favors the Mahomes-era Kansas City Chiefs, coinciding with the team’s emergence as a key driver of TV viewership/ratings and, thereby, revenue. Our study suggests that financial reliance on dominant entities can alter enforcement dynamics, a concern with implications far beyond sports governance.
That is from a new piece by Spencer Barnes, Ted Dischman, and Brandon Mendez. Via the excellent Kevin Lewis.
Sentences to ponder
By ordering the U.S. military to summarily kill a group of people aboard what he said was a drug-smuggling boat, President Trump used the military in a way that had no clear legal precedent or basis, according to specialists in the laws of war and executive power.
Mr. Trump is claiming the power to shift maritime counterdrug efforts from law enforcement rules to wartime rules. The police arrest criminal suspects for prosecution and cannot instead simply gun suspects down, except in rare circumstances where they pose an imminent threat to someone.
Here is more from the NYT.
The polity that is Brazil
Yet perhaps the biggest reason spending is high is that the constitution requires it. The charter mandates an extraordinary 90% of all federal spending. Notably it ties most public pensions to wage growth, and requires health and education spending to rise in line with revenue growth. If Brazil were to end most tax exemptions and undo these two policies, its debt-to-gdp ratio, which is already above 90%, would be almost 20 percentage points lower by 2034 than it would be without any reform, reckons the IMF. To deal with all this, what is really needed is to amend the constitution.
High spending and a tangle of subsidised credit schemes also reduce the effectiveness of monetary policy. That means the central bank must increase rates even higher to control inflation. Brazil’s real interest rate of 10% is among the highest in the world. Such rates cripple investment and drag down growth, while well-connected businessmen can get their hands on artificially cheap rates.
Among those who must pay the full rate is the government itself.
And:
Tax exemptions total 7% of gdp, up from 2% in 2003 (see chart 2). Dozens of sectors receive tax breaks or credit subsidies on the basis that they are national champions, or from “temporary” help that has never ended. Brazil’s courts cost 1.3% of gdp, making them the second-most expensive in the world, with much of that going on cushy pensions and perks. Some $15bn a year, or 78% of the military budget, is spent on pensions and salaries. The United States spends just one-quarter of the defence budget on personnel.
Here is more from The Economist.
Inside India’s endless trials
The FT’s Krishn Kaushik covers the courts in India:
…in one recent example a Delhi court concluded a property dispute after 66 years. Both the original litigants were dead. Still, the lawyer for one of the warring parties cautioned that the conclusion was in fact not the end, as the ruling would be appealed.
Three years ago, after pondering a dispute for 16 years, the supreme court sent back a 60-year-old land case for fresh adjudication to a lower court, which had already taken over 30 years to give its judgment in 2006.
A 2021 study [excellent study, AT] of Mumbai real estate found that more than a quarter of the projects under planning or construction and 43 per cent of all “built-up spaces” in the city were under some litigation. My apartment block was one of them.
…One of the reasons for this accumulation is human resources. India has around 16 judges per million people, compared to over 150 for the US. In 2016, the issue brought the country’s chief justice, TS Thakur, to tears during a speech as he requested that the government hire more judges to wade through the “avalanche” of backlog.
Reminds me of one of my favorite MR posts, A Twisted Tale of Rent Control in the Maximum City.
*Adolescence*
This is a British TV show, in four episodes, available on Netflix. It is the first TV show I have wanted to watch in about four years. I cannot review it without immediate spoilers, but I can tell you it takes place in a school, in a criminal justice system, and in a family. Northern England, so do turn on those subtitles. Here are some reviews.