Category: Medicine
The microfoundations of the baby boom?
Between 1936 and 1957, fertility rates in the U.S. increased 62 percent and the maternal mortality rate declined by 93 percent. We explore the effects of changes in maternal mortality rates on white and nonwhite fertility rates during this period, exploiting contemporaneous or lagged changes in maternal mortality at the state-by-year level. We estimate that declines in maternal mortality explain 47-73 percent of the increase in fertility between 1939 and 1957 among white women and 64-88 percent of the increase in fertility among nonwhite women during our sample period.
Here is the full article by Christopher Handy and Katharine Shester, via the excellent Kevin Lewis. Overall, I take this as a negative for the prospect of another, future baby boom? We just cannot make maternity all that much safer, starting from current margins.
Privatizing Law Enforcement: The Economics of Whistleblowing
The False Claims Act lets whistleblowers sue private firms on behalf of the federal government. In exchange for uncovering fraud and bringing the case, whistleblowers can receive up to 30% of any recovered funds. My work on bounty hunters made me appreciate the idea of private incentives in the service of public goals but a recent paper by Jetson Leder-Luis quantifies the value of the False Claims Act.
Leder-Luis looks at Medicare fraud. Because the government depends heavily on medical providers to accurately report the services they deliver, Medicare is vulnerable to misbilling. It helps, therefore, to have an insider willing to spill the beans. Moreover, the amounts involved are very large giving whistleblowers strong incentives. One notable case, for example, involved manipulating cost reports in order to receive extra payments for “outliers,” unusually expensive patients.
On November 4, 2002, Tenet Healthcare, a large investor-owned hospital company, was sued under the False Claims Act for manipulating its cost reports in order to illicitly receive additional outlier payments. This lawsuit was settled in June 2006, with Tenet paying $788 million to resolve these allegations without admission of guilt.
The savings from the defendants alone were significant but Leder-Luis looks for the deterrent effect—the reduction in fraud beyond the firms directly penalized. He finds that after the Tenet case, outlier payments fell sharply relative to comparable categories, even at hospitals that were never sued.
Tenet settled the outlier case for $788 million, but outlier payments were around $500 million per month at the time of the lawsuit and declined by more than half following litigation. This indicates that outlier payment manipulation was widespread… for controls, I consider the other broad types of payments made by Medicare that are of comparable scale, including durable medical equipment, home health care, hospice care, nursing care, and disproportionate share payments for hospitals that serve many low-income patients.
…the five-year discounted deterrence measurement for the outlier payments computed is $17.46 billion, which is roughly nineten times the total settlement value of the outlier whistleblowing lawsuits of $923 million.
[Overall]…I analyze four case studies for which whistleblowers recovered $1.9 billion in federal funds. I estimate that these lawsuits generated $18.9 billion in specific deterrence effects. In contrast, public costs for all lawsuits filed in 2018 amounted to less than $108.5 million, and total whistleblower payouts for all cases since 1986 have totaled $4.29 billion. Just the few large whistleblowing cases I analyze have more than paid for the public costs of the entire whistleblowing program over its life span, indicating a very high return on investment to the FCA.
As an aside, Leder-Luis uses synthetic control but allows the controls to come from different time periods. I’m less enthused by the method because it introduces another free parameter but given the large gains at small cost from the False Claims Act, I don’t doubt the conclusion:
The results of this analysis suggest that privatization is a highly effective way to combat fraud. Whistleblowing and private enforcement have strong deterrence effects and relatively low costs, overcoming the limited incentives for government-conducted antifraud enforcement. A major benefit of the False Claims Act is not just the information provided by the whistleblower but also the profit motive it provides for whistleblowers to root out fraud.
Are the ACA exchanges unraveling?
After all, that is what economists predicted if the mandate was not tightly enforced. Here is the latest reprt:
Premiums for the most popular types of plans sold on the federal health insurance marketplace Healthcare.gov will spike on average by 30 percent next year, according to final rates approved by the Centers for Medicare and Medicaid Services and shown in documents reviewed by The Washington Post.
The higher prices — affecting up to 17 million Americans who buy coverage on the federal marketplace — reflect the largest annual premium increases by far in recent years.
Here is the full article.
The MR Podcast: Our Favorite Models, Session 2: The Baumol Effect
On The Marginal Revolution Podcast this week we continue discussing some of our favorite models with a whole episode on the Baumol effect (with a sideline into the Linder effect). I say our favorite models, but the Baumol Effect is not one of Tyler’s favorite models! I thought this was a funny section:
TABARROK: When you look at all of these multiple sectors, the repair sector, repairing of clothing, repairing of shoes, repairing of cars, repairing of people, it’s not an accident that these are all the same thing. Healthcare is the repairing of people. Repair services, in general, have gone up because it’s a very labor-intensive area of the economy. It’s all the same thing. That’s why I like the Baumol effect, because it explains a very wide set of phenomena.
COWEN: A lot of things are easier to repair than they used to be, just to be clear. You just buy a new one.
TABARROK: That’s my point. You just buy a new one.
COWEN: It’s so cheap to buy a new one.
TABARROK: Exactly. The new one is manufactured. That’s the whole point, is the new one takes a lot less labor. The repair is much more labor intensive than the actual production of the good. When you actually produce the good, it’s on a factory floor, and you’ve got robots, and they’re all going through da-da-da-da-da-da-da. Repair services, it’s unique.
COWEN: I think you’re not being subjectivist enough in terms of how you define the service. The service for me, if my CD player breaks, is getting a stream of music again. That is much easier now and cheaper than it used to be. If you define the service as the repair, well, okay, you’re ruling out a lot of technological progress. You can think of just diversity of sources of music as a very close substitute for this narrow vision of repair. Again, from the consumer’s point of view, productivity on “repair” has been phenomenal.
TABARROK: That is a consequence of the Baumol effect, not a denial of the Baumol effect. Because of the Baumol effect, repair becomes much more expensive over time, so people look for substitutes. Yes, we have substituted into producing new goods. It works both ways. The new goods are becoming cheaper to manufacture. We are less interested in repair. Repair is becoming more expensive. We’re more interested in the new goods. That’s a consequence of the Baumol effect.
You can’t just say, “Oh, look, we solved the repair problem by throwing things out. Now we don’t have to worry about repairs.” Yes, that’s because repair became so much more expensive. A shift in relative prices caused people to innovate. I’m not saying that innovation doesn’t happen. One of the reasons that innovation happens is because the relative price of repair services is going up.
COWEN: That’s a minor effect. It’s not the case that, oh, I started listening to YouTube because it became too expensive to repair my CD player. It might be a very modest effect. Mostly, there’s technological progress. YouTube, Spotify, and other services come along, Amazon one-day delivery, whatever else. For the thing consumers care about, which is never what Baumol wanted to talk about. He always wanted to fixate on the physical properties of the goods, like the anti-Austrian he was.
It’s just like, oh, there’s been a lot of progress. It takes the form of networks with very complex capital and labor interactions. It’s very hard to even tease out what is truly capital intensive, truly labor intensive. You see this with the AI companies, all very mixed together. That just is another way of looking at why the predictions are so hard. You can only get the prediction simple by focusing very simply on these nonsubjectivist, noneconomic, physical notions of what the good has to be.
TABARROK: I think there’s too much mood affiliation there, Tyler.
COWEN: There’s not enough Kelvin Lancaster in Baumol.
Here’s the episode. Subscribe now to take a small step toward a much better world: Apple Podcasts | Spotify | YouTube.
The unraveling of Obamacare?
Paul Krugman has a recent post defending the exchange subsidies and tax credits that the Republicans wish to cut, talking with Jonathan Cohn about the “premium apocalypse” (and here). Whether or not one agrees with Krugman normatively, the arguments if anything convince me that Obamacare probably is not financially or politically stable.
To recap some history briefly:
1. Prior to passage, ACA advocates assured us that all three “legs of the stool” were necessary, most of all the mandate, to prevent adverse selection and skyrocketing premia. That argument made sense and was accepted by most economists, whether or not they favored ACA.
2. Obamacare passes by razor-thin margins, with a mandate.
3. The mandate proves extremely unpopular. Whether or not it is efficient, it puts a disproportionate share of the cost burden on other policy purchasers through the exchanges. The Republicans run against ACA and make some big gains.
4. Trump in essence “saves” Obamacare by in essence defusing enforcement of the mandate. The people who hated paying the very high premia could now back out of the system without getting into real trouble. As a result, much of the opposition to Obamacare, and the scare stories about expensive policies, dissipates.
5. Contrary to the predictions of the economists, Obamacare does not collapse. Enough people kept on signing up, perhaps because there is often a fair degree of “positive selection” into insurance coverage. Still, one has to wonder whether this will last.
6. Under the Biden administration, the Democrats support the continuation of premium support, but not with massive enthusiasm. It is expensive, though of course the Democrats did understand this is a centerpiece of Obamacare and they cannot give up on it. If you are calling the current situation a “premium apocalypse,” a lot of money has to be involved.
7. Putting aside the current Trump plans and the government shutdown and concomitant fight, how stable is this budget allocation over time? Is it possible that the economists (including Krugman and David Cutler) were right all along, albeit with a long lag, and the exchanges ultimately cannot work without a mandate? And that the premium support will just get more and more expensive?
It seems to be this scenario, while hardly proven, is really quite possible. One can blame Trump of course, but maybe the allocation no longer is sustainable over the medium term?
During the ACA debates, Megan McArdle frequently made the point that such a big policy passed by such small margins could not so easily last. A lot of people wanted to look past that observation, but was she so wrong?
Addendum: By the way, how are we supposed to pay for all of this? Repealing the recent Trump tax cuts and raising taxes on the rich doesn’t seem to come close to bringing the budget into balance. Endorse a VAT if you wish, but then do so! And let us have that debate. In the meantime everyone is just playing games with us.
More corruption in the Harvard leadership
Harvard School of Public Health Dean Andrea A. Baccarelli received at least $150,000 to testify against Tylenol’s manufacturer in 2023 — two years before he published research used by the Trump administration to link the drug to autism, a connection experts say is tenuous at best.
Baccarelli served as an expert witness on behalf of parents and guardians of children suing Johnson & Johnson, the manufacturer of Tylenol at the time. U.S. District Court Judge Denise L. Cote dismissed the case last year due to a lack of scientific evidence, throwing out Baccarelli’s testimony in the process.
“He cherry-picked and misrepresented study results and refused to acknowledge the role of genetics in the etiology” of autism spectrum disorder or ADHD, Cote wrote in her decision, which the plaintiffs have since appealed.
Here is more from The Crimson.
AI and the FDA
Dean Ball has an excellent survey of the AI landscape and policy that includes this:
The speed of drug development will increase within a few years, and we will see headlines along the lines of “10 New Computationally Validated Drugs Discovered by One Company This Week,” probably toward the last quarter of the decade. But no American will feel those benefits, because the Food and Drug Administration’s approval backlog will be at record highs. A prominent, Silicon Valley-based pharmaceutical startup will threaten to move to a friendlier jurisdiction such as the United Arab Emirates, and they may in fact do it.
Eventually, I expect the FDA and other regulators to do something to break the logjam. It is likely to perceived as reckless by many, including virtually everyone in the opposite party of whomever holds the White House at the time it happens. What medicines you consume could take on a techno-political valence.
Agreed—but the nearer-term upside is repurposing. Once a drug has been FDA approved for one use, physicians can prescribe it for any use. New uses for old drugs are often discovered, so the off-label market is large. The key advantage of off-label prescribing is speed: a new use can be described in the medical literature and physicians can start applying that knowledge immediately, without the cost and delay of new FDA trials. When the RECOVERY trial provided evidence that an already-approved drug, dexamethasone, was effective against some stages of COVID, for example, physicians started prescribing it within hours. If dexamethasone had had to go through new FDA-efficacy trials a million people would likely have died in the interim. With thousands of already approved drugs there is a significant opportunity for AI to discover new uses for old drugs. Remember, every side-effect is potentially a main effect for a different condition.
On Ball’s main point, I agree: there is considerable room for AI-discovered drugs, and this will strain the current FDA system. The challenge is threefold.
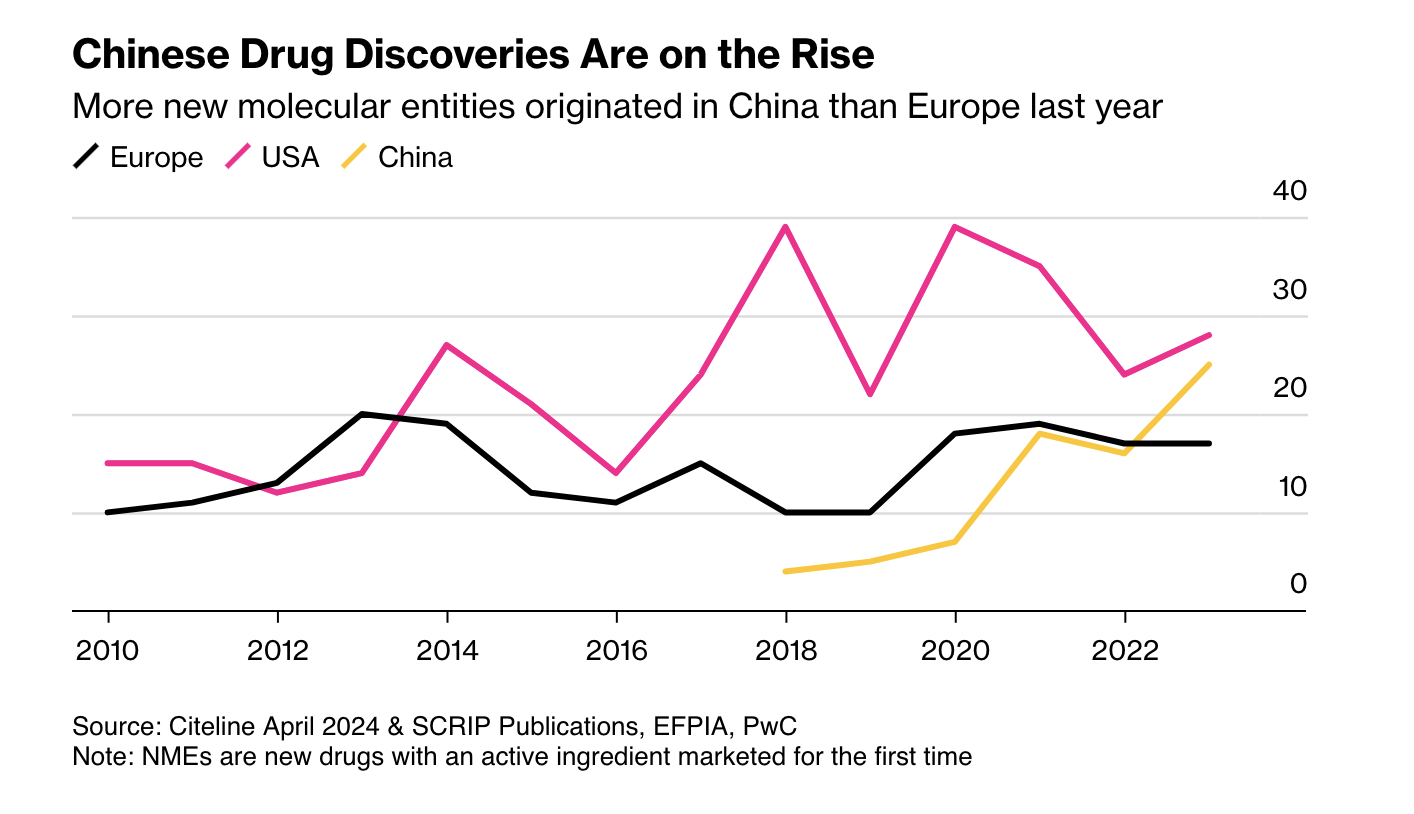
First, as Ball notes, more candidate drugs at lower cost means other regulators may become competitive with the FDA. China is the obvious case: it is now large and wealthy enough to be an independent market, and its regulators have streamlined approvals and improved clinical trials. More new drugs now emerge from China than from Europe.
Second, AI pushes us toward rational drug design. RCTs were a major advance, but they are in some sense primitive. Once a mechanic has diagnosed a problem, the mechanic doesn’t run a RCT to determine the solution. The mechanic fixes the problem! As our knowledge of the body grows, medicine should look more like car repair: precise, targeted, and not reliant on averages.
Closely related is the rise of personalized medicine. As I wrote in A New FDA for the Age of Personalized, Molecular Medicine:
Each patient is a unique, dynamic system and at the molecular level diseases are heterogeneous even when symptoms are not. In just the last few years we have expanded breast cancer into first four and now ten different types of cancer and the subdivision is likely to continue as knowledge expands. Match heterogeneous patients against heterogeneous diseases and the result is a high dimension system that cannot be well navigated with expensive, randomized controlled trials. As a result, the FDA ends up throwing out many drugs that could do good.
RCTs tell us about average treatment effects, but the more we treat patients as unique, the less relevant those averages become.
AI holds a lot of promise for more effective, better targeted drugs but the full promise will only be unlocked if the FDA also adapts.
Girls improve student mental health
Using individual-level data from the Add Health surveys, we leverage idiosyncratic variation in gender composition across cohorts within the same school to examine whether being exposed to a higher share of female peers affects mental health and school satisfaction. We find that being exposed to a higher proportion of female peers, despite only improving school satisfaction for boys, improves mental health for both boys and girls. The benefits are greater among boys of low socioeconomic backgrounds, who would otherwise be more likely to be exposed to violent and disruptive peers. We find suggestive evidence that the mechanisms driving our findings are consistent with stronger school friendships for boys and better self-image and grades for girls.
That is from a new NBER working paper by Monica Deza and Maria Zhu.
Is it the phones?
Or perhaps we should just credit Sydney Sweeney? That is from Chris Said.
One look at negative emotional contagion
This paper studies how peers’ genetic predisposition to depression affects own mental health during adolescence and early adulthood using data from the National Longitudinal Study of Adolescent to Adult Health (Add Health). I exploit variation within schools and across grades in same-gender grademates’ average polygenic score—a linear index of genetic variants—for major depressive disorder (the MDD score). An increase in peers’ genetic risk for depression has immediate negative impacts on own mental health. A one standard deviation increase in same-gender grademates’ average MDD score significantly increases the probability of being depressed by 1.9 and 3.8 percentage points for adolescent girls (a 7.2% increase) and boys (a 25% increase), respectively. The effects persist into adulthood for females, but not males. I explore several potential mechanisms underlying the effects and find that an increase in peers’ genetic risk for depression in adolescence worsens friendship, increases substance use, and leads to lower socioeconomic status. These effects are stronger for females than males. Overall, the results suggest that there are important social-genetic effects in the context of mental health.
That is from a recent paper by Yeongmi Jeong, via the excellent Kevin Lewis.
Where are the trillion dollar biotech companies?
In today’s market, even companies with multiple approved drugs can trade below their cash balances. Given this, it is truly perplexing to see AI-biotechs raise mega-rounds at the preclinical stage – Xaira with a billion-dollar seed, Isomorphic with $600M, EvolutionaryScale with $142M, and InceptiveBio with $100M, to name a few. The scale and stage of these rounds reflect some investors’ belief that AI-biology pairing can bend the drug discovery economics I described before.
To me, the question of whether AI will be helpful in drug discovery is not as interesting as the question of whether AI can turn a 2-billion-dollar drug development into a 200-million-dollar drug development, or whether 10 years to approve a drug can become 5 years to approve a drug. AI will be used to assist drug discovery in the same way software has been used for decades, and, given enough time, we know it will change everything [4]. But is “enough time” 3 years or half a century?
One number that is worth appreciating is that 80% of all costs associated with bringing a drug to market come from clinical-stage work. That is, if we ever get to molecules designed and preclinically validated in under 1 year, we’ll be impacting only a small fraction of what makes drug discovery hard. This productivity gain cap is especially striking given that the majority of the data we can use to train models today is still preclinical, and, in most cases, even pre-animal. A perfect model predictive of in vitro tox saves you time on running in vitro tox (which is less than a few weeks anyway!), doesn’t bridge the in vitro to animal translation gap, and especially does not affect the dreaded animal-to-human jump. As such, perfecting predictive validity for preclinical work is the current best-case scenario for the industry. Though we don’t have a sufficient amount and types of data to solve even that.
Here is the full and very interesting essay, from the excellent Lada Nuzhna.
Decker and KingoftheCoast on single payer health insurance
Here is the conclusion of the piece:
To reiterate, the key point in this piece is that high administrative costs in US healthcare are unlikely to represent “do-nothing waste.” Some of the purported costs are entirely fake. To include them in the possible savings of single payer shows either ignorance or dishonesty. Some of the costs are to prevent waste and fraud, which should be paid by Medicare now (although they are not). Of what is left, the cost of duplication pales in comparison to the plausible benefits of choice and competition in health insurance. When you put all of these together, the case for single-payer is nonexistent. A better system would be to subsidize those who are too poor to pay, scrap the government health insurance providers and the VA, remove the employer tax deduction, and allow providers to compete.
Here is the full piece, excellent work.
Do not forget
Estimating real-world vaccine effectiveness is vital to assessing the coronavirus disease 2019 (COVID-19) vaccination program and informing the ongoing policy response. However, estimating vaccine effectiveness using observational data is inherently challenging because of the nonrandomized design and potential for unmeasured confounding. We used a regression discontinuity design to estimate vaccine effectiveness against COVID-19 mortality in England using the fact that people aged 80 years or older were prioritized for the vaccine rollout. The prioritization led to a large discrepancy in vaccination rates among people aged 80–84 years compared with those aged 75–79 at the beginning of the vaccination campaign. We found a corresponding difference in COVID-19 mortality but not in non-COVID-19 mortality, suggesting that our approach appropriately addressed the issue of unmeasured confounding factors. Our results suggest that the first vaccine dose reduced the risk of COVID-19 death by 52.6% (95% confidence limits: 15.7, 73.4) in those aged 80 years, supporting existing evidence that a first dose of a COVID-19 vaccine had a strong protective effect against COVID-19 mortality in older adults. The regression discontinuity model’s estimate of vaccine effectiveness is only slightly lower than those of previously published studies using different methods, suggesting that these estimates are unlikely to be substantially affected by unmeasured confounding factors.
From Charlotte Bermingham, et.al. There is plenty of other research yielding broadly similar conclusions. The Covid vaccines saved millions of lives, well over two million lives even from a conservative estimate.
For the pointer I thank Alex T.
Dean Ball on state-level AI laws
He is now out of government and has resumed writing his Substack. Here is one excerpt from his latest:
Several states have banned (see also “regulated,” “put guardrails on” for the polite phraseology) the use of AI for mental health services. Nevada, for example, passed a law (AB 406) that bans schools from “[using] artificial intelligence to perform the functions and duties of a school counselor, school psychologist, or school social worker,” though it indicates that such human employees are free to use AI in the performance of their work provided that they comply with school policies for the use of AI. Some school districts, no doubt, will end up making policies that effectively ban any AI use at all by those employees. If the law stopped here, I’d be fine with it; not supportive, not hopeful about the likely outcomes, but fine nonetheless.
But the Nevada law, and a similar law passed in Illinois, goes further than that. They also impose regulations on AI developers, stating that it is illegal for them to explicitly or implicitly claim of their models that (quoting from the Nevada law):
(a) The artificial intelligence system is capable of providing professional mental or behavioral health care;
(b) A user of the artificial intelligence system may interact with any feature of the artificial intelligence system which simulates human conversation in order to obtain professional mental or behavioral health care; or
(c) The artificial intelligence system, or any component, feature, avatar or embodiment of the artificial intelligence system is a provider of mental or behavioral health care, a therapist, a clinical therapist, a counselor, a psychiatrist, a doctor or any other term commonly used to refer to a provider of professional mental health or behavioral health care.
First there is the fact that the law uses an extremely broad definition of AI that covers a huge swath of modern software. This means that it may become trickier to market older machine learning-based systems that have been used in the provision of mental healthcare, for instance in the detection psychological stress, dementia, intoxication, epilepsy, intellectual disability, or substance abuse (all conditions explicitly included in Nevada’s statutory definition of mental health).
But there is something deeper here, too. Nevada AB 406, and its similar companion in Illinois, deal with AI in mental healthcare by simply pretending it does not exist. “Sure, AI may be a useful tool for organizing information,” these legislators seem to be saying, “but only a human could ever do mental healthcare.”
And then there are hundreds of thousands, if not millions, of Americans who use chatbots for something that resembles mental healthcare every day. Should those people be using language models in this way? If they cannot afford a therapist, is it better that they talk to a low-cost chatbot, or no one at all? Up to what point of mental distress? What should or could the developers of language models do to ensure that their products do the right thing in mental health-related contexts? What is the right thing to do?
The State of Nevada would prefer not to think about such issues. Instead, they want to deny that they are issues in the first place and instead insist that school employees and occupationally licensed human professionals are the only parties capable of providing mental healthcare services (I wonder what interest groups drove the passage of this law?).
Free the Patient: A Competitive-Federalism Fix for Telemedicine
During the pandemic, many restrictions on telemedicine were lifted, making it far easier for physicians to treat patients across state lines. That window has largely closed. Today, unless a doctor is separately licensed in a patient’s state—or the states have a formal agreement—remote care is often illegal. So if you live in Virginia and want a second opinion from a Mayo Clinic physician in Florida, you may have to fly to Florida, unless that Florida physician happens to hold a Virginia license.
The standard framing says this is a problem of physician licensing. That leads directly to calls for interstate compacts or federalizing medical licensure. Mutual recognition is good. Driver’s licenses are issued by states but are valid in every state. No one complains that Florida’s regime endangers Virginians. But mutual recognition or federal licensing is not the only solution nor the only way to think about this issue.
The real issue isn’t who licenses doctors. It’s that patients are forbidden from choosing a licensed doctor in another state. We can keep state-level licensing, but free the patient. Let any American consult any physician licensed in any state. That’s competitive federalism—no compacts, no federal agency, just patient choice.
A close parallel comes from credit markets. After Marquette Nat. Bank v. First of Omaha (1978), host states could no longer block their residents from using credit cards issued by national banks chartered elsewhere. A Virginian can legally borrow on a South Dakota credit card at South Dakota’s rates. Nothing changed about South Dakota’s licensing; what changed was the prohibition on choice.
Consider Justice Brennan’s argument in this case:
“Minnesota residents were always free to visit Nebraska and receive loans in that state.” It hadn’t been suggested that Minnesota’s laws would apply in that instance, he added. Therefore, they shouldn’t be applied just because “the convenience of modern mail” allowed Minnesotans to get credit without having to visit Nebraska.
Exactly analogously, everyone agrees that Virginia residents are free to visit Florida and be treated by Florida physicians. No one suggests that Virginia’s laws should follow VA residents to Florida. Therefore, VA’s laws shouldn’t be applied just because the convenience of modern online tools allow Virginians to get medical advice and consultation without having to visit Florida.
In short, patients should be allowed to choose physicians as easily as borrowers choose banks.
